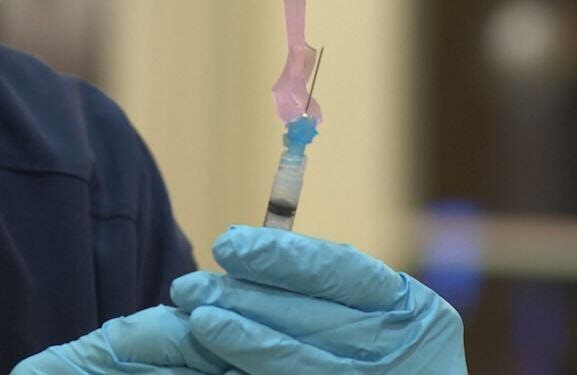
MARYLAND-A Maryland-based company, Novavax, is hoping to get FDA approval for another version of the COVID-19 vaccine as they are now entering Phase 3 of their trials. Novavax has reportedly received $1.6 billion from the federal government and aims to enroll about 30,000 volunteers to test the vaccine both in the U.S. and in Mexico.
Unlike Pfizer’s and Moderna’s messenger RNA-based vaccines, Novavax President of Research and Development, Dr. Gregory Glenn, explained that their technology uses so-called “spike” proteins.
"We have a very good expression, that we would call it a 3D printer, where we put the gene in, we make the protein, we take that protein and that's in our vaccine," Dr. Glenn said. "Now people get immunity to the spike protein and when they see the virus, that immunity will block the binding, and they're completely protected."
At the moment, there are three main types of COVID vaccines, mRNA, which is Pfizer and Moderna, viral vector, which is what the UK AstraZeneca uses, and then there’s Novavax's protein vaccine, which is an older technology. They all require patients get two dosis of the vaccine.

"Their vaccine will actually just inject little pieces of that antigen, as opposed to the Pfizer and Moderna having our bodies build the antigen, they will actually inject that antigen, not the whole virus, they are not giving a whole virus in a vaccine, you cannot get sick from the Novavax, meaning you cannot get infected with COVID--with any of those vaccines we talked about," explained Dr. Bill Chasanov, the COVID-19 medical director at Beebe Healthcare.
The company said it’s enrolling a diverse participant body, which would include a 10 to 20% Latinx and a 15% African-American population. At least a fourth of their participants must also be ages 65 or older.
Although at this point in time, Phase 3 does not guarantee FDA approval, Dr. Glenn said their advancements from Phase 1 and 2 trials show promising results.
"The products that are being deployed right now require extra logistics because they need to be kept very, very cold," Dr. Glenn added. "That's not a characteristic from our vaccine, our vaccine is stable, it can be distributed in refrigeration. You can see that, although we've got vaccines that have been approved now, getting them out to the public is really a big challenge, and also just the magnitude of the number of people that need to be immunized...so there's a lot of interest in our vaccine because of that."
Dr. Chasanov said it’s important to remind ourselves that the pandemic is still ongoing and recommends everyone follow the 'three Ws" to stay safe.
"Washing your hands and wearing a mask and watching your distance are still the three Ws that are extremely important if you receive the vaccine, or you don’t receive the vaccine or you’re waiting for the vaccine," he said.
Dr. Chasanov also explained that herd immunity generally exists when at least 60 to 80 percent of a given population receive the vaccine, so some leniency in social distancing for example, could happen perhaps by the end of the summer.
Doctors across Delmarva told WRDE that they are urging everyone to get vaccinated, regardless of the brand itself as soon as it is available to them, and those with autoimmune diseases should ask their infectious specialist doctor to seriously consider getting vaccinated because the mild secondary effects (such as a possible fever), outweigh the effects of contracting the Coronavirus.